
THE FACE OF FUSION
ACDF | ALIF | LLIF | PLIF | TLIF
HEDRON™ 3D printed titanium interbody spacers feature a biomimetic porous scaffold designed to promote bone formation onto and through the implant.



TRABECULAR BONE
INSPIRED DESIGN
Osseointegration between an implant and surrounding bone may help achieve stability. HEDRON™ integrates biomimetic architecture with characteristics of established interbody fusion devices.
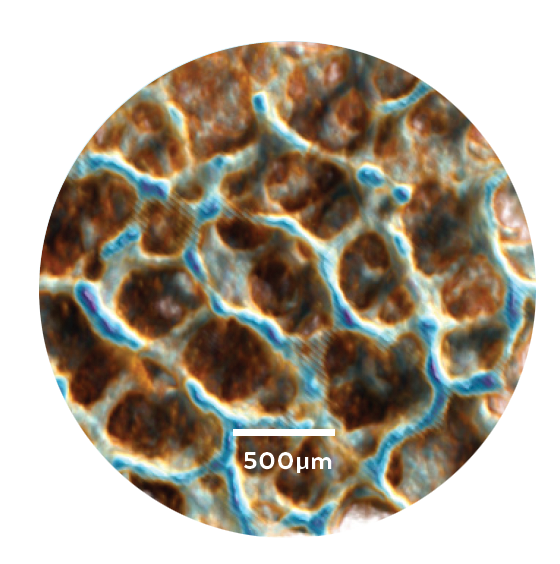
MicroCT of trabecular bone

SEM image of HEDRON™
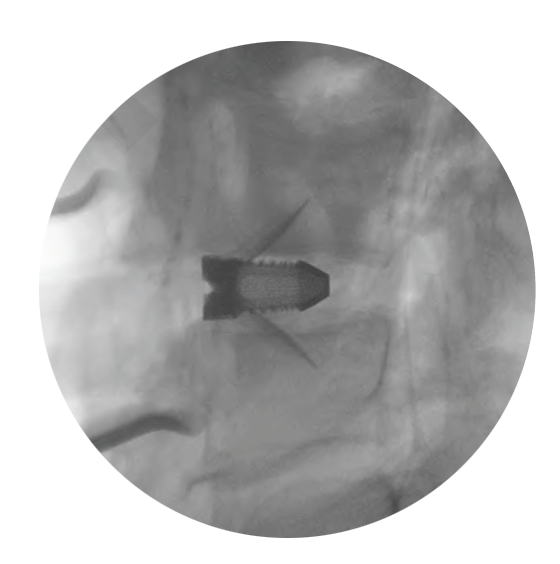
Clear visualization
Supplemental fixation required.
FEATURES
Lattice stiffness comparable to trabecular bone1
70% porosity
Expansive pore size distribution
Roughened surface texture
1. Mechanical study data on file.
Strength and Porosity
Unlike first generation 3D printed implants (grid and open architecture), HEDRON™ strikes the ideal balance of strength and porosity through a sturdy frame and pore size distribution similar to trabecular bone.
Grid
Open architecture
HEDRON™
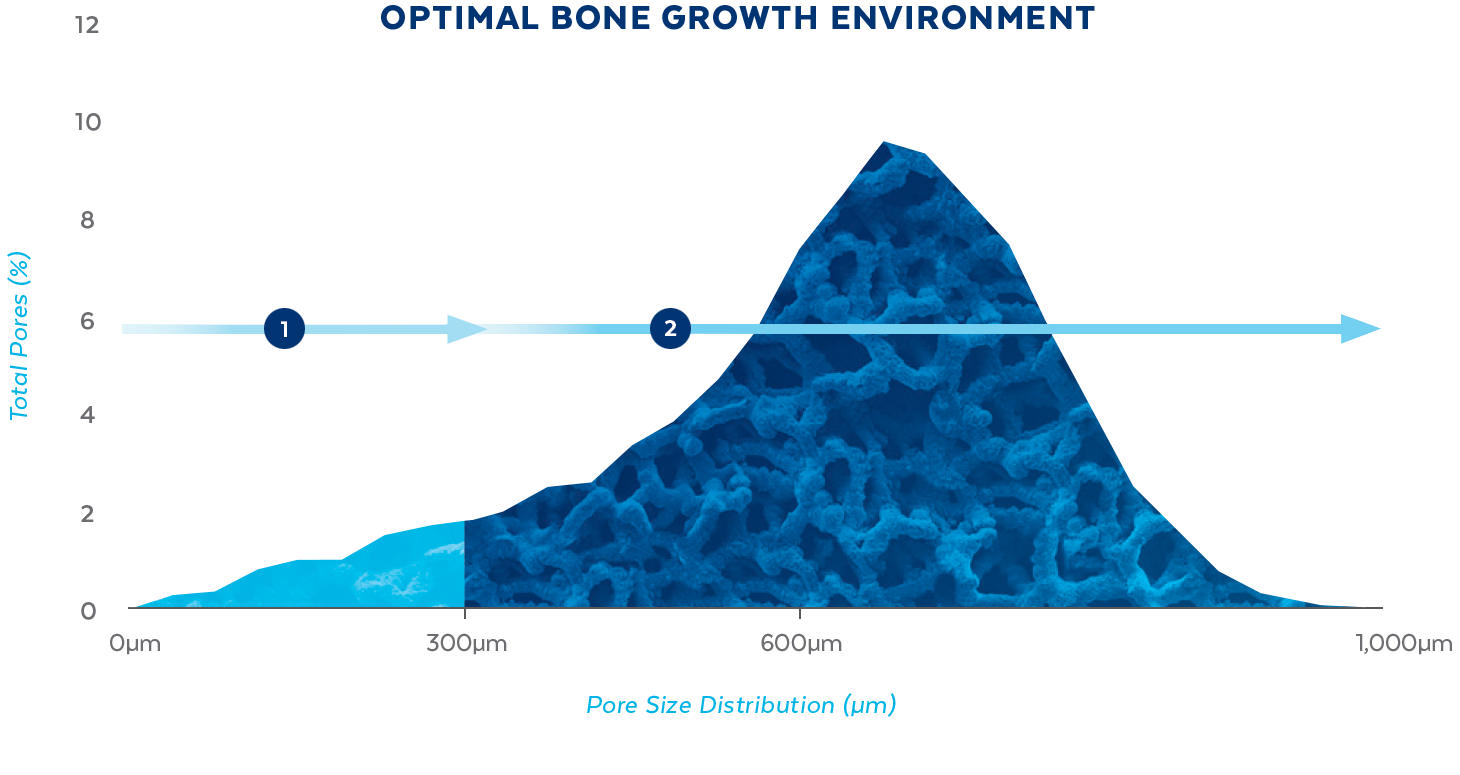

ONGROWTH
<300μm
Small pores (<300μm) are recommended to support initial surface adhesion2

BONE FORMATION
>300μm
Large pores (>300μm) have been shown to lead to direct osteogenesis3
2. Torres-Sanchez et al. Material Science and Engineering. 2017 Mar; 219–228.
3. KKarageorgiou V, Kaplan D. Biomaterials. 2005;26(27):5474-91.
ENCOURAGES CELLULAR
RESPONSE
In Vitro testing demonstrated that HEDRON™ generated a greater expression of Vascular Endothelial Growth Factor (VEGF) and Osteocalcin, two biological indicators of bone formation when seeded with osteoblasts.4
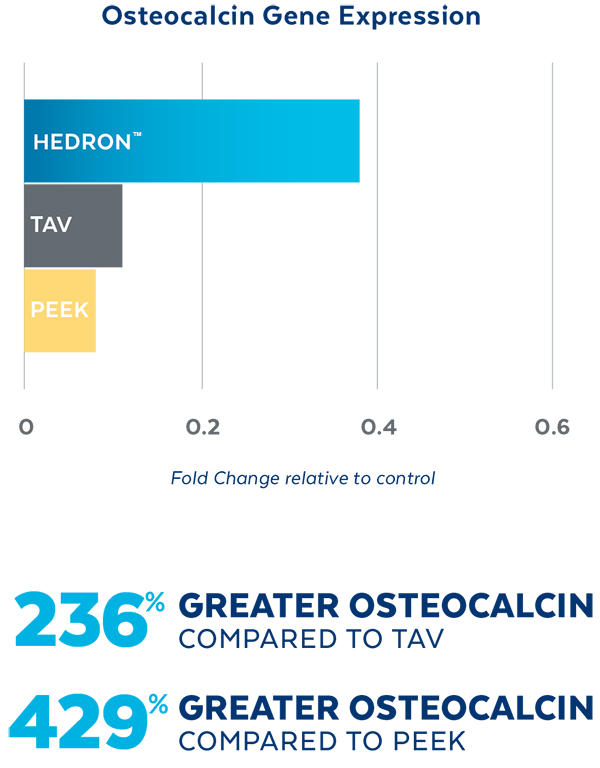
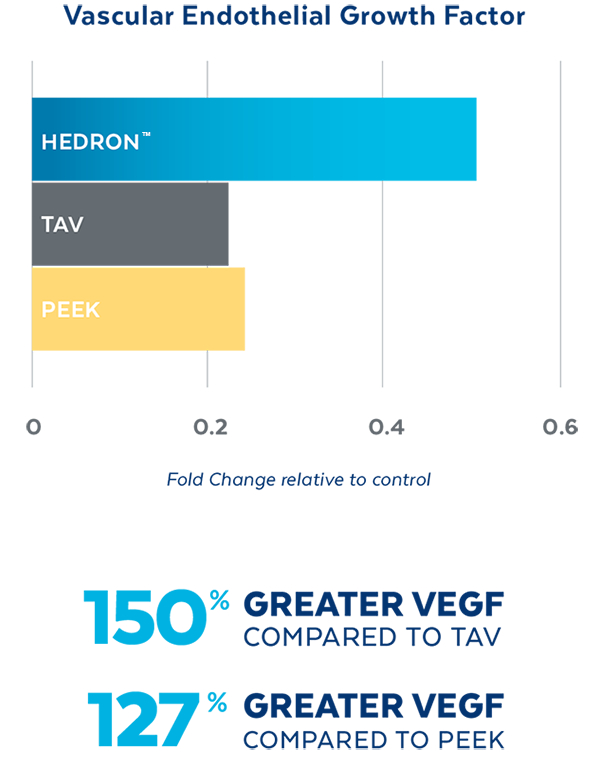
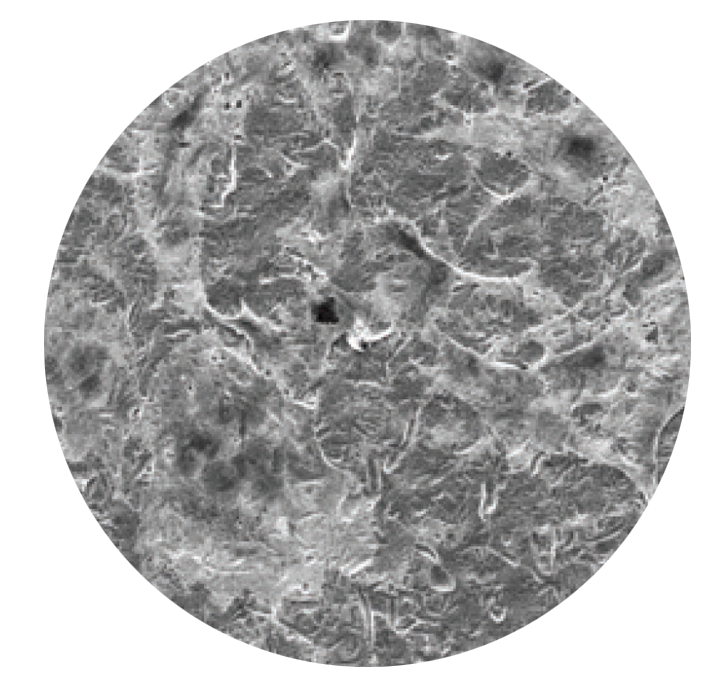
SEM images of cell proliferation at 21 days (500x magnification)

PEEK
TAV
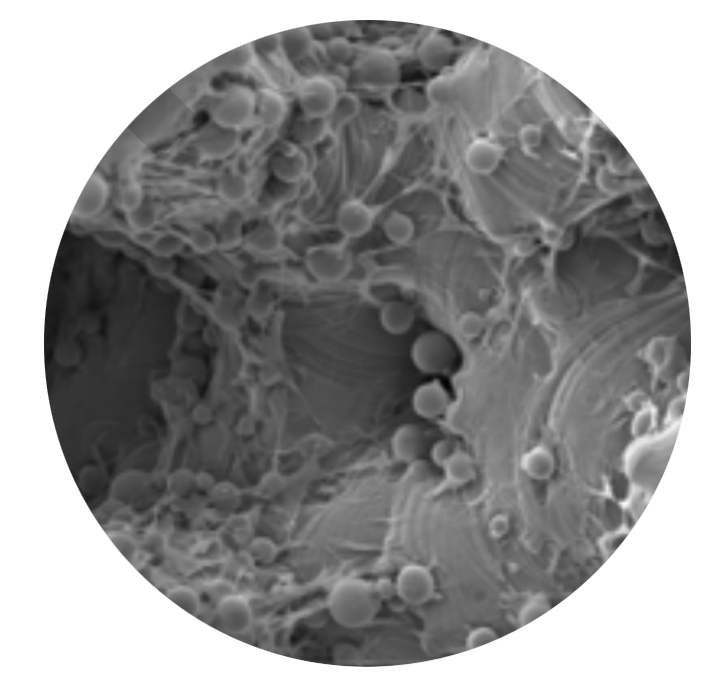
HEDRON™
4. Cell study data on file.
PROMOTES BONE
FORMATION
Unlike PEEK and TAV, the porous architecture of HEDRON™ allows for bone to grow through the spacer walls and incorporate into the fusion mass. In a pre-clinical ovine study, HEDRON™ implants showed significantly more bone growth compared to PEEK and titanium implants at 6-weeks post-op.5
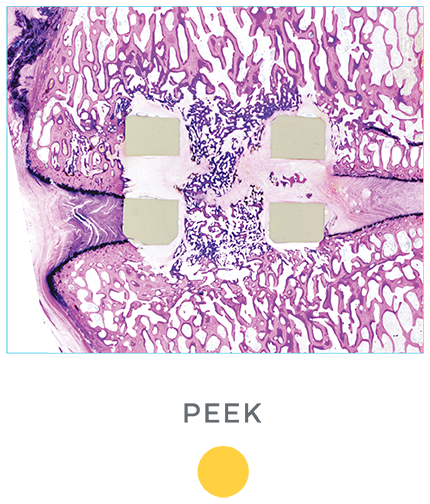
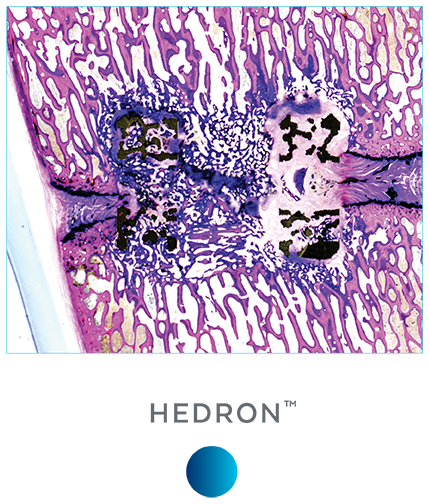
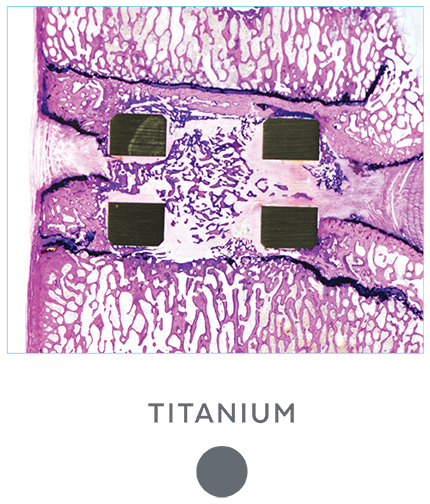
A pre-clinical sheep interbody study using HEDRON™, Titanium (Ti), and PEEK implants was performed to compare bone ingrowth and ongrowth.

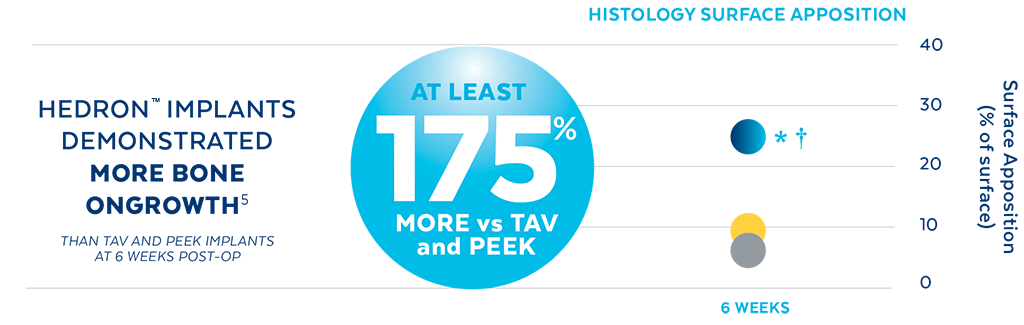
5. Animal study data on file.
*p<0.05 vs PEEK.
†p<0.05 vs TAV
Accommodates Multiple Approaches and Techniques
| Procedure | HEDRON™ Spacer | Footprint | Heights | Lordosis |
| ACDF | 
HEDRON C™ |
12x14, 14x16, 15x18mm | 5, 6, 7, 8, 9, 10, 11, 12mm | 0°, 7°, 12°, 15°, 20° |
 HEDRON IC™ |
12x14, 14x16, 15x18mm | 5, 6, 7, 8, 9, 10, 11, 12mm | 0°, 7°, 12° | |
| ALIF |  HEDRON A™ |
22x29, 24x35, 28x39mm | 9, 11, 13, 15, 17, 19, 21mm | 8°, 15°, and 20° |
 HEDRON IA™ |
24x30, 26x34, 29x39mm | 11, 13, 15, 17, 19, 21mm | 8°, 15°, 20°, 25°, and 30° | |
| LLIF |  HEDRON L™ |
18, 22mm widths 40–60mm heights | 7, 9, 11, 13, 15mm | 10°, 15° |
| PLIF |  HEDRON P™ |
8x22, 10x22, 10x26, 10x30, 12x26, 12x30mm | 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17mm | 8°, 15° |
| TLIF |  HEDRON T™ |
10x28, 11x33mm | 7, 8, 9, 10, 11, 12, 13, 15, 17mm | 8°, 15° |
